TL;DR Science: The Greenhouse Gases
By Angelo Chen
March 16, 2022 · 6 minute read
Earth Science
Environmental Science
As climate change worsens, sea levels rise, and global temperatures increase, greenhouse gases have become a common topic of discussion. But what are they really? What makes them greenhouse gases, and how could this knowledge be useful in reducing their influence on the greenhouse effect? This week, I’ll be answering all of these questions in part 2 of a 3-part series on the greenhouse effect and greenhouse gases. Before reading, we recommend that you check out part 1 in last month’s article here!
What They Are
By definition, a greenhouse gas is any gas that contributes to the greenhouse effect by absorbing infrared radiation. As we know, although we cannot see infrared radiation, we can feel it in the form of heat. Greenhouse gases contribute to the greenhouse effect by absorbing this heat, and releasing it back toward the Earth.
With an understanding of what the greenhouse effect is, we can now dive into what all of the greenhouse gases are, and how they compare. Specifically, the most prominent greenhouse gases are water vapor (H2O), carbon dioxide (CO2), methane (CH4), ozone (O3), nitrous oxide (N2O), and fluorinated gases.
The effects of greenhouse gases are typically measured in two ways: how long they live in the atmosphere, as well as how severe their influence is. The former is simply measured in years, and the latter is measured using the Global Warming Potential (GWP). This value is a direct comparison between each gas and carbon dioxide, as CO2 was used as the basis for this value. The GWP of each gas is found by taking the ratio between the amount of energy 1 ton of a greenhouse gas can absorb in 100 years and the amount of energy 1 ton of carbon dioxide can absorb in 100 years.
Water Vapor

The largest contributor to the greenhouse effect is water vapor, the gaseous form of water. H2O, like carbon dioxide, has similar chemical properties in that it can absorb infrared radiation and radiate the heat back towards Earth. However, the relative concentration of water vapor in Earth’s atmosphere is regulated by the water cycle, thus making human regulation of water vapor in our atmosphere unrealistic.
For comparison with the other gases, water vapor has one of the shortest lifespans, being just one or two weeks. It also has a low GWP of roughly 0.0005 to 0.001. These numbers make water vapor seem like a very weak greenhouse gas; it would be, if not for its massive concentration in our atmosphere. The sheer amount of water vapor in the atmosphere well makes up for its weakness; water vapor contributes to roughly 50% of the greenhouse effect we observe today.
Carbon Dioxide
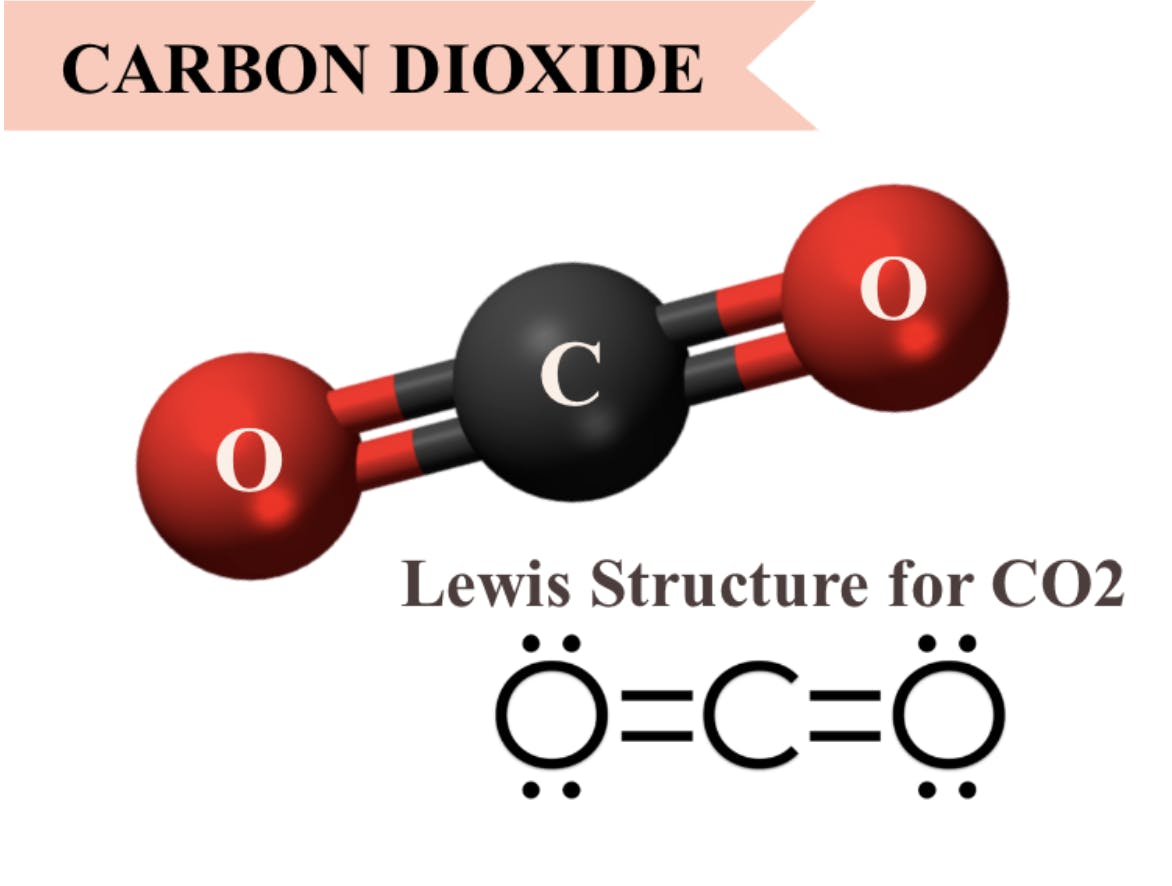
Perhaps the most commonly known greenhouse gas is carbon dioxide. Unlike water vapor, the concentration of carbon dioxide in our atmosphere is heavily impacted by human activities, which is why many are calling for reforms in carbon emissions. It is also capable of absorbing infrared radiation that other greenhouse gases, like water, cannot. Specifically, CO2 absorbs wavelengths of 15 microns very well, which H2O does not. Carbon dioxide also cannot absorb smaller wavelengths, which allows things like ultraviolet radiation to penetrate into the Earth’s atmosphere, but doesn’t allow the re-emitted infrared radiation to escape.
One of the reasons why CO2 and other greenhouse gases are so good at absorbing infrared radiation has to do with their molecular formula. Gases with three or more molecules are often capable of absorbing infrared radiation because they can vibrate in multiple ways, unlike diatomic molecules like O2 and N2. This is the same mechanism that allows each of the other greenhouse gases to absorb infrared radiation as well.
For more information, check out the animation here on how carbon dioxide and other polyatomic molecules vibrate in ways that allow them to interact with infrared radiation!
Molecules Vibrate | Center for Science Education
By definition, carbon dioxide has a GWP of 1, because it is used as the basis for the GWP value. One of the main reasons why carbon dioxide emissions are hotly debated is because of how long it lasts in our atmosphere. Once added, CO2 can last anywhere from 300 to 1000 years. This means that once the worst effects of global warming start to manifest, it will be too late for us to decrease carbon emissions, as the amount of carbon dioxide already in the atmosphere will not decrease very quickly.
Methane
Methane is another greenhouse gas that is commonly discussed. For the same reason as carbon dioxide, CH4 is also capable of absorbing infrared radiation, contributing to the greenhouse effect in the same way. Methane has a GWP of roughly 28-36, but only stays in the atmosphere for 12 years, not nearly as long as carbon dioxide.
This may make it seem like methane is a lesser issue than carbon dioxide, but this is untrue. The GWP measure is created with a 100 year expectation. However, since methane only lasts 12 years, this value is diluting the 12 year effect of methane across a 100 year period. Indeed, using a 20-year GWP measure, methane’s GWP value increases to roughly 84 - 87.
Ozone
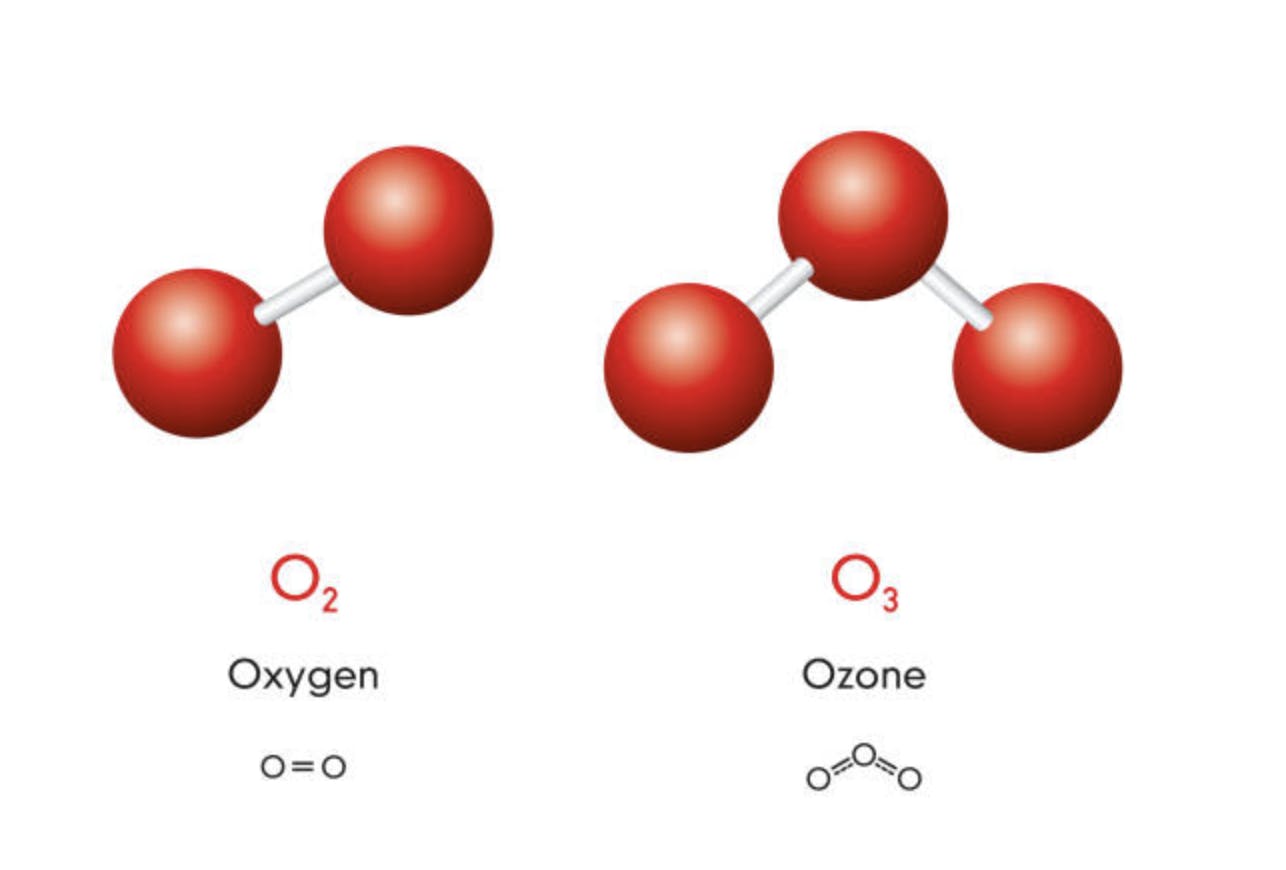
Ozone is an interesting gas, because it can either be beneficial or harmful, depending on where it is in our atmosphere. Higher up in our atmosphere, it creates the ozone layer, which protects the Earth from harmful ultraviolet radiation. But in lower elevations, it does contribute to the greenhouse effect in the same way carbon dioxide does. Nonetheless, its beneficial effect far outweighs its negative effects, so regulation of ozone emissions are of little concern.
Nitrous Oxide
Nitrous oxide is a gas that is commonly overlooked when discussing greenhouse gases. Its function is similar to that of carbon dioxide, but with a significantly worse effect. It has a GWP of 298, and lasts about 114 years in the atmosphere. Even after accounting for the fact that it doesn’t last as long as carbon dioxide, its influence on our atmosphere is far greater than that of carbon dioxide, which is why restrictions on nitrous oxide emissions are being set in place for many sectors, like agriculture and forestry.
Fluorinated Gases
Fluorinated gases are characterized by their composition, containing fluorine, as well as their similar effects on the atmosphere. Fluorinated gases have the highest GWP measures among any of the greenhouse gases previously listed. According to 2019 statistics on these gases, the GWP values of several fluorinated gases are as follows:
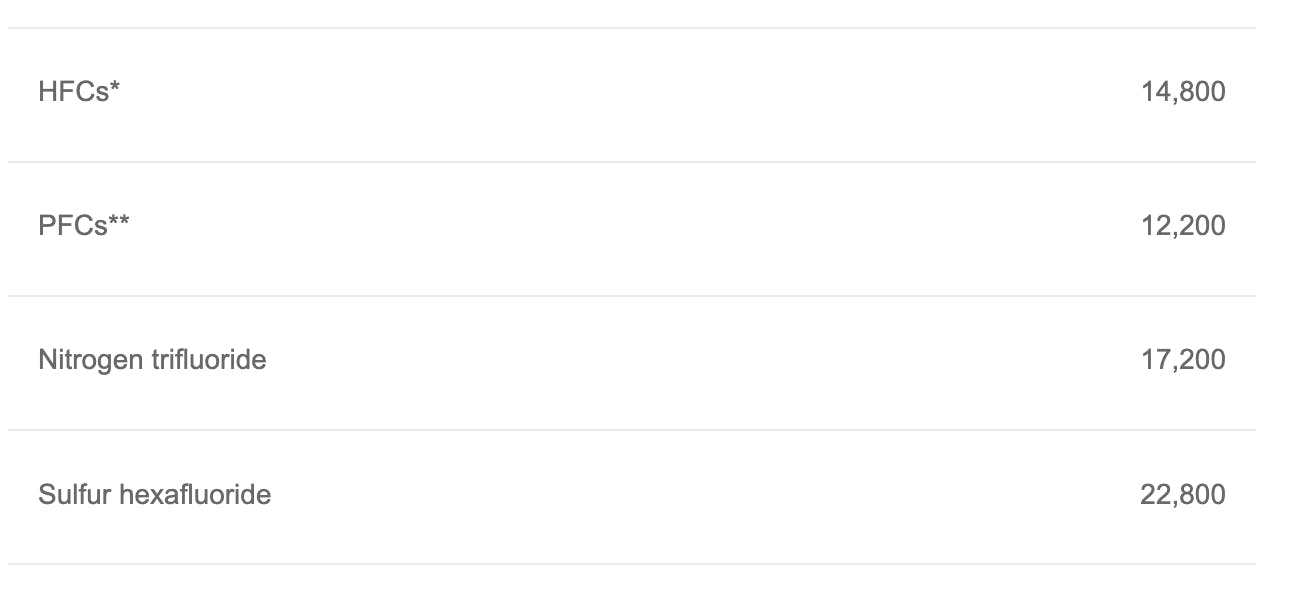
That’s in the tens of thousands! Furthermore, their effects are exemplified by their long lifespans as well. Sulfur hexafluoride (SF6), with a GWP of 22,800, also lasts around 3,200 years. It is immediately obvious why these gases are of great concern.
In addition to their potency as greenhouse gases, they also can have a variety of other negative effects on the atmosphere as well. Take chlorofluorocarbons, or CFCs, as an example. These gases were used in the manufacturing of aerosols, but their effect on the ozone layer caused so much damage that an international treaty, the Montreal Protocol, was created to help save the ozone layer.
TL;DR:
There are many greenhouse gases, of which the main ones are water vapor (H2O), carbon dioxide (CO2), methane (CH4), ozone (O3), nitrous oxide (N2O), and fluorinated gases. These are all capable of trapping infrared radiation in the atmosphere contributing to the greenhouse effect. Their influences differ widely in how much energy they hold, and how long they last in the atmosphere, but they all give rise to the climate change and global warming we see today.
Sources
https://climate.mit.edu/ask-mit/how-do-greenhouse-gases-trap-heat-atmosphere
https://iopscience.iop.org/article/10.1088/1748-9326/aae018/meta
https://www.epa.gov/ghgemissions/overview-greenhouse-gases
https://www.epa.gov/ghgemissions/understanding-global-warming-potentials
https://clear.ucdavis.edu/explainers/why-methane-cattle-warms-climate-differently-co2-fossil-fuels
https://www.eia.gov/tools/faqs/faq.php?id=84&t=11
https://energyeducation.ca/encyclopedia/Greenhouse_gas
https://www.statista.com/statistics/1085616/global-warming-potential-fluorinated-gases/
Image Sources
https://www.bioexplorer.net/greenhouse-gases.html/
https://interestingengineering.com/water-vapor-could-be-surprise-source-of-renewable-energy
https://youaskweanswer.net/is-carbon-dioxideco2-polar-or-nonpolar/
Did you enjoy this article?
About The Author
Angelo is currently a junior at Suncoast Community High School. He enjoys spending time with his dog, and playing volleyball and violin as hobbies. He also enjoys doing math, in and out of school. If you have any questions about this article, feel free to contact him at angelo@sciteens.org.